Preparing for an Audit
Preparing for an audit is a very important step for your organization. The external audit from a 3rd party registrar is the final step before your organization receives certification to ISO 13485.
The auditors have several objectives:
- Verify that your documentation meets all of the requirements of the standard
- Ensure your employees know their role in the QMS and are familiar with the ISO 13485 requirements that pertain to their role in the organization.
- The procedures and processes are correctly followed.
When the day of the registration audit has arrived, it is time to showcase your QMS and how you have addressed the requirements. Being audited is time-consuming and costly so you want to make sure that you are well prepared. Here are some tips from 13485Store.com to help your organization ensure that you are ready in order to have a successful audit.
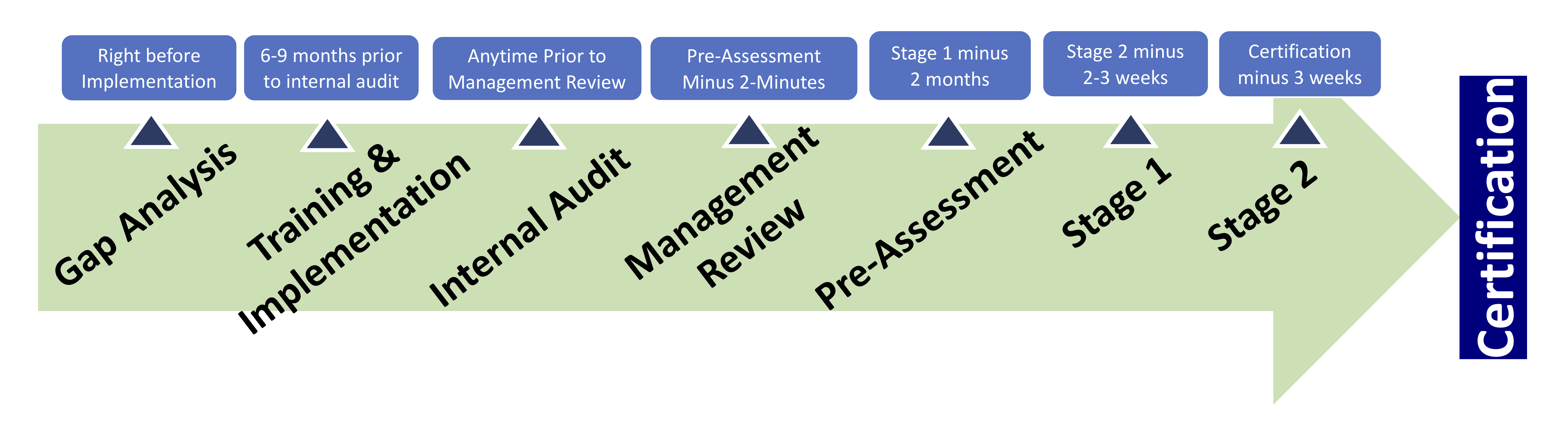
Give the organization ample time to prepare
Preparing your organization for an audit takes time and effort. When you have decided that receiving certification is necessary for the organization, you should set goals and give an appropriate amount of time to meet the requirements of the standard.
Prepare the employees
Your employees are the ones that will demonstrate that your organization is complying with the requirements of ISO 13485. Before the audit you will want to make sure that all employees know when the audit will be taking place and what the scope of the audit is. Each employee should have a clear idea of the organization’s quality objectives, and how their role contributes to them. Each employee should have proper training on the tasks that they perform.
When you are preparing your employees to answer questions, remember this is not a test, auditors are not looking for an exact answer, rather they want to know how employees go about finding information and answers. Do procedures provide information? If the information is not in a procedure, where does the employee go next, do they ask a supervisor?
13485Store.com suggests Employee Training Courses to help prepare your organization for the audit:
Documented Information
Documented information is another important aspect of preparing for your audit. When you are preparing for your audit, you need to ensure that documents and record lists have been updated. Additionally, all documents need to have been reviewed, approved, communicated and followed by everyone involved in the process or activity. You should also ensure that no one is using outdated documents.
Processes and Procedures
Processes and Procedures need to be carefully followed. Your organization needs to make sure all processes are meeting the planned arrangement you set out to achieve, and that they are being followed and performed in the same and correct way by all employees.
Prepare the facility
When you are preparing for the audit your organization needs to be well-organized and cleaned. If your facility is a mess, it is easy to miss nonconformances. You want to make sure that you are well aware of every area of the facility so that you do not have to encounter any hidden issues during the audit. When getting your organized ready for the audit, check bulletin boards, counters, drawers, etc. for uncontrolled documents, uncalibrated measuring and monitoring instruments and unidentified parts or supplies.
Internal Audit
The organization must conduct an internal audit. Not only is this a requirement of the ISO standard, but it is also the best way to catch any major nonconformances. Internal audits should be taken seriously. They are also a great way to prepare staff for the interview process. Remember that the purpose of an internal audit is to identify weaknesses and areas that need improvement in order to increase the effectiveness of the management system and prepare for external audits.
13485Store.com offers the following products to help your organization perform a successful internal audit:
Management Review
The management review should take place following the internal audit. This step should consist of reviewing past internal audit findings and take corrective actions where necessary.
Review past Internal Audit Findings
After your organization undergoes an Internal Audit, these findings should be reviewed in a closing meeting with everyone involved. The purpose of an internal audit is to identify possible weaknesses and areas that need to be improved in order to better the management system and meet the requirements of ISO 13485, and earn certification. To have a successful audit, it is a must that the process of the audit is carried out with both auditors and auditees.
The meeting should be detailed to give the organization an understanding of where they are falling short and how they can improve. This information should be given in an objective and friendly manner and suggestions should be informed constructively.
Review Corrective Action Process
Reviewing past audits and taking corrective action to any nonconformances is key to certification. We recommend taking corrective actions immediately to avoid compromising your ability to achieve certification. A major problem found by the auditor could delay your certification and leave you scrambling to establish a process to fix the issue. Performing internal audits in preparation for your third-party auditor is ideal.
Pre-Assessment
A pre-assessment audit is performed with the same independence and objectivity as a 2-stage certification audit. The auditor will conduct an assessment that monitors documentation review, process review, etc. in order to understand where the organization is relative to meeting the standard’s requirements.
Pre-assessment audits are performed on-site and are a complete assessment of the ISO 13485 quality management system. All nonconformities and observations found will be presented in an audit report that will be delivered at the end of the process. This report will allow the organization to improve its processes and implement any necessary corrective actions.
Pre-assessment audits are recommended for any organization that has implemented a management system and wishes to undergo a certification audit. A Pre-Assessment audit can be conducted by qualified consultants, registrars, or other competent individuals that understand the management system being audited. The organization that will perform the 2-stage certification audit may also offer pre-assessment audits as well.
2-Stage Certification Audit
After your organization has completed the above steps you will be ready for the 2-stage certification audit. Learn more about the 2-stage certification audit.
Have a professional audit
When the auditor arrives, your goal should be making a good first impression and to act professionally. Auditors are there to help the organization uncover any nonconformances and weaknesses in order to take action, improve and become certified. Remember they want your organization to succeed as well.
Related Articles:


